Local Contact Person for Pharmacovigilance
Automating LCPPV tasks across the globe
Before Tepsivo entered the field in 2020,
all available LCPPV services were far from efficiency
The work of local safety officers or local QPPVs around the world was highly uncoordinated and requiring a big effort from so called “project managers” to keep things somewhat together.
such processes came with
insanely high costs
induced large number of
nonsense hours
and ultimately led to
risks to legal compliance
No provider showed real effort in automating this aspect of PV.
We saw the gap. And soon fixed this.
Industry standard of LCPPV management

Tepsivo LCPPV Network

Industry standard of LCPPV management

Tepsivo LCPPV Network
No provider showed real effort in automating this aspect of PV.
We saw the gap. And soon fixed this.

Industry standard of LCPPV management

Tepsivo LCPPV Network
No provider showed real effort in automating this aspect of PV.
We saw the gap. And soon fixed this.
The Tepsivo LCPPV Network led the charge
in our complete automation of pharmacovigilance
across the global PV system for our customers.
One central place
to manage all local PV
On a daily basis, we maintain an active network of LCPPVs spanning 150+ countries, already having supported over 50 pharma companies in the first 5 years since company conception.
We’ve done this without ever invoicing a single cent in made-up charges such as “project management”.
How is this all possible?
Thanks to the unique system of Tepsivo OnePV, where we have eliminated all redundant admin tasks.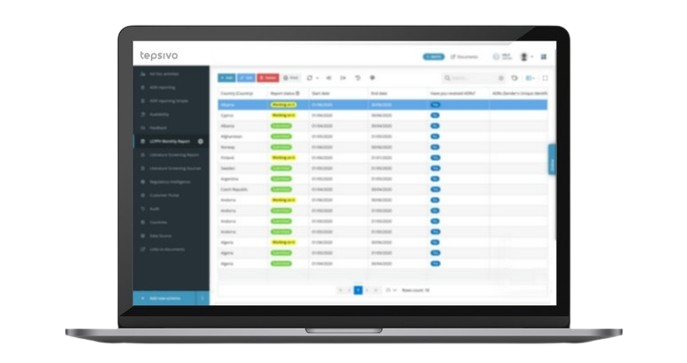
Full automation effectively means
LCPPVs focusing on scince, not admin
We have also taken away routine local PV tasks such as local literature monitoring and regulatory intelligence which most companies assign to LCPPVs.
These are fully automated within Tepsivo OnePV and its modules ‘Tepsivo Literature’ and ‘Tepsivo News’.
Tepsivo Literature
Available through ‘Literature Review’ tab in Tepsivo OnePV, this module automatically lists articles from countless local literature sources.
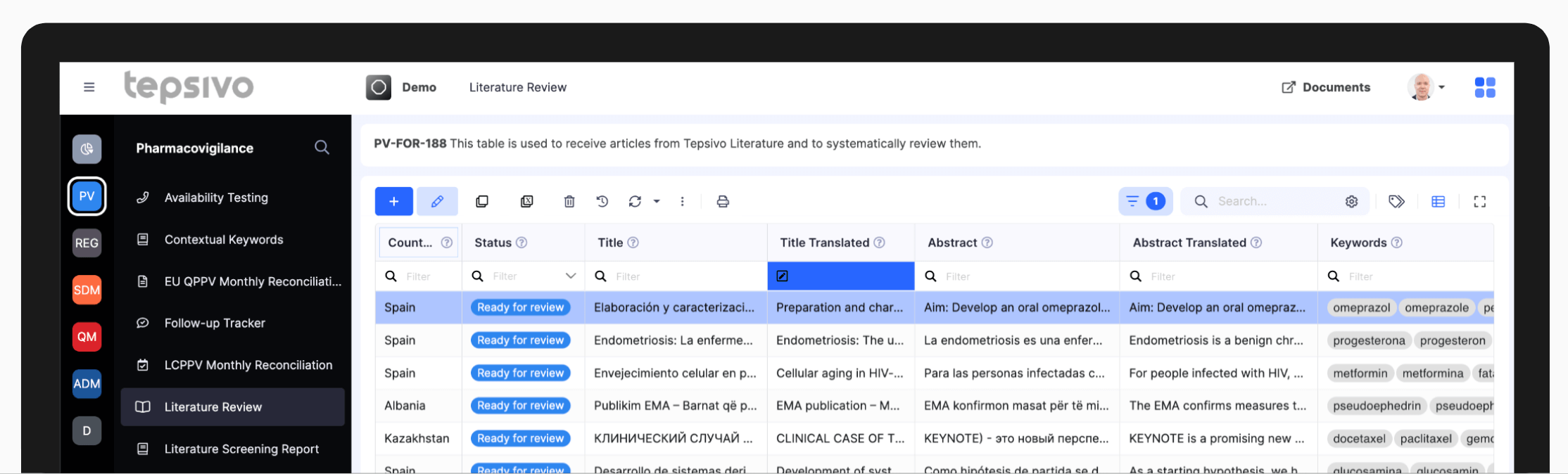
Tepsivo Literature
Available through ‘Literature Review’ tab in Tepsivo OnePV, this module automatically lists articles from countless local literature sources.
Tepsivo Literature
Available through ‘Literature Review’ tab in Tepsivo OnePV, this module automatically lists articles from countless local literature sources.

100%
legal compliance
2 LCPPVs per country
at minimum
600 EUR
per month / country all inclusive
By taking away mundane administrative tasks, LCPPVs have time for value-added activities. This also means we can afford to hire the most qualified experts, not having to race to the bottom on costs just to stay competitive.
Tepsivo LCPPV network is unmatched in expertise, compliance, and cost. Not to speak of the further benefits of complete integration within one PV system.
What more
would you need?
A safety report from an LCPPV
in a safety database and at the authority within minutes?
Perfectly possible within Tepsivo OnePV and its integrated safety reporting from original case intake through to authority submission and back for signal analysis.
Tepsivo Safety Database
A safety report can be quickly sent to the Tepsivo Safety Database, part of Tepsivo OnePV.
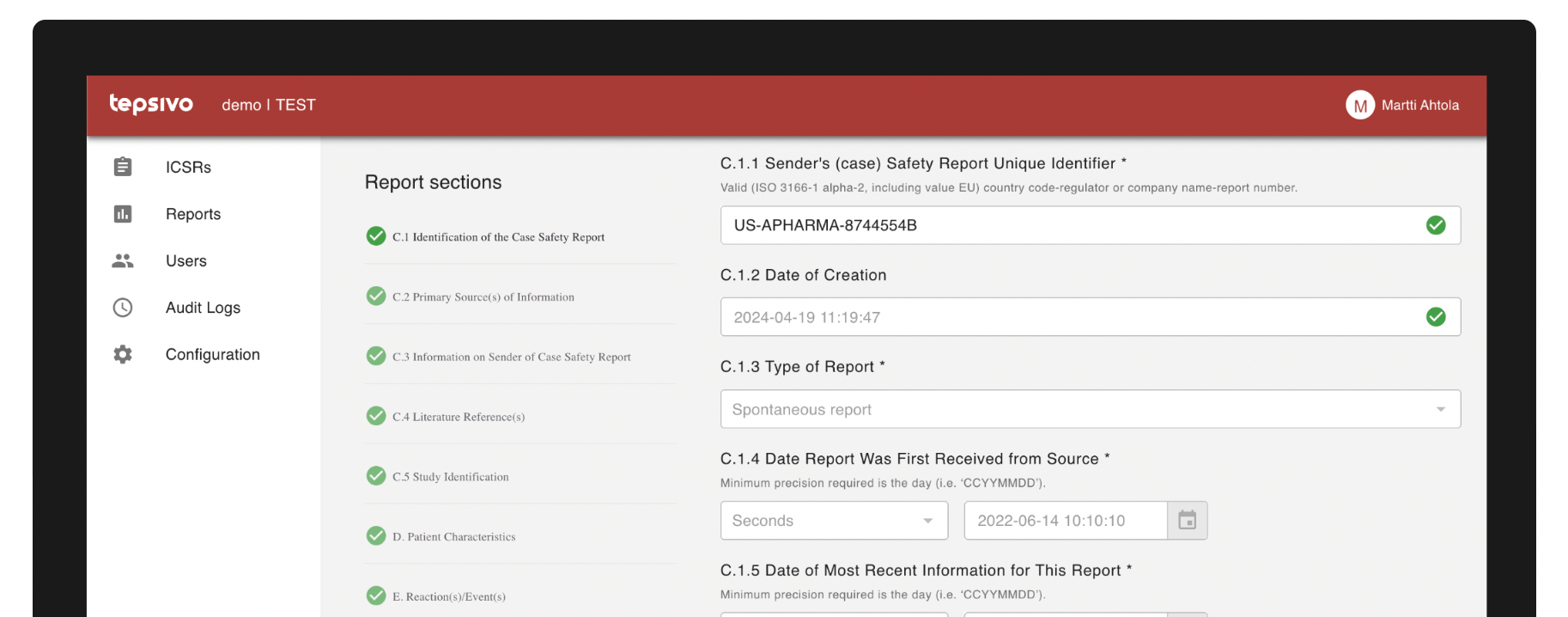
Tepsivo Safety Database
A safety report can be quickly sent to the Tepsivo Safety Database, part of Tepsivo OnePV.
Tepsivo Safety Database
A safety report can be quickly sent to the Tepsivo Safety Database, part of Tepsivo OnePV.

Complete 24/7 oversight of all local PV
and affiliate activities around the world in one tool?
Exactly what a central integrated system Tepsivo OnePV enables, including both completely automated audit trail and reconciliation.
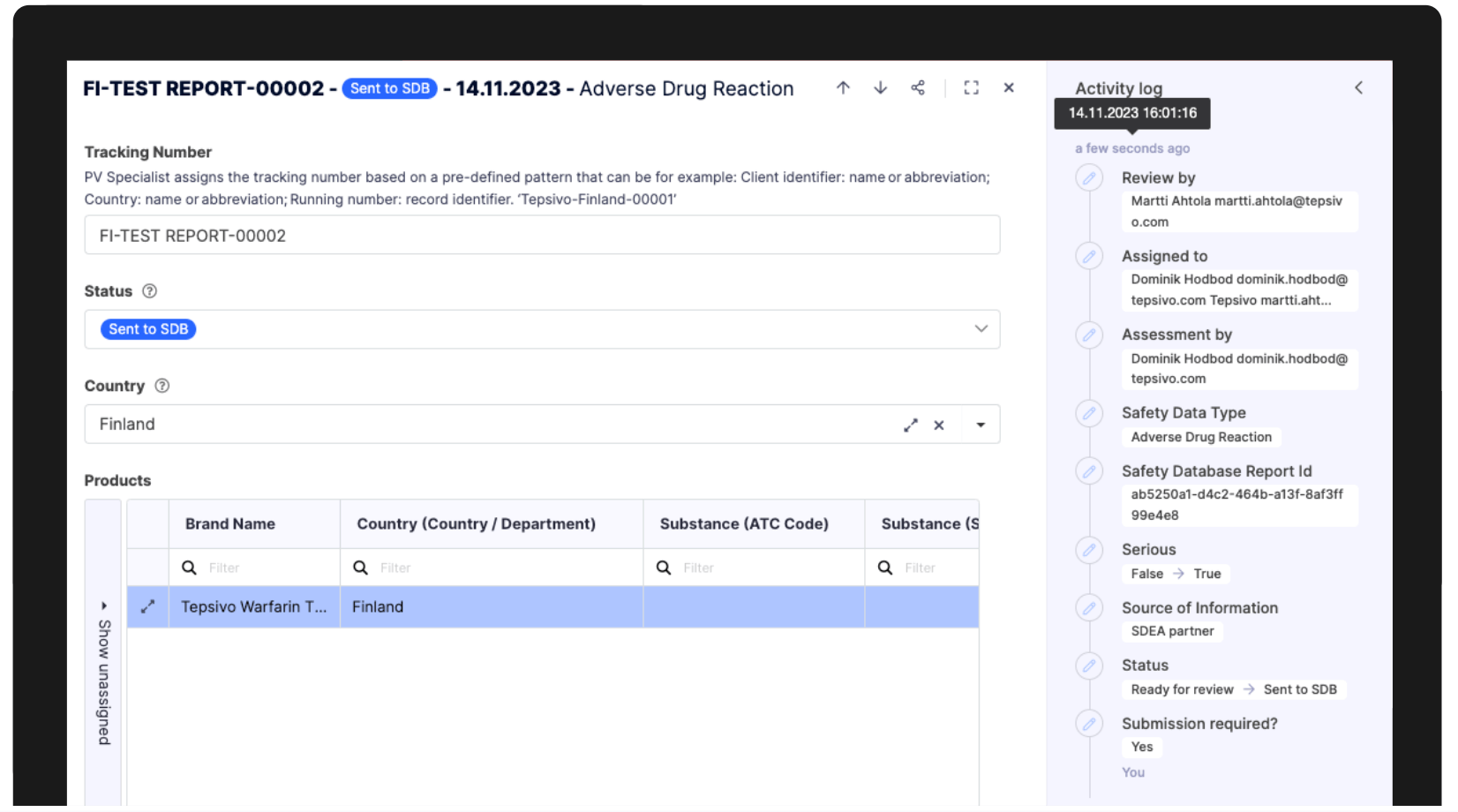
Exactly what a central integrated system Tepsivo OnePV enables, including both completely automated audit trail and reconciliation.

Activity log
On the level of a single record, every step and change of the processes is tracked.
This information is also available across all records in ‘Audit trail’ section of Tepsivo OnePV solution.

150+
countries covered
2 weeks
to-go live date at max.
100%
automation of typical
routine tasks

40+
happy customers globally
zero
project management charges
one
central place to manage
all your PV activities
150+
countries covered
2 weeks
to-go live date
at max.
100%
automation of typical
routine tasks

40+
happy customers globally
zero
project management charges
one
central place to manage
all your PV activities
Let’s have a chat
Whatever your needs are, we look forward to getting in touch with you.
Feel free to drop us a message and we will contact you right away.
Tepsivo Oy | Haartmaninkatu 4, Building 14, 00290, Helsinki, Finland | VAT number FI31367614 | contact@tepsivo.com | +358 402 204 698 | Privacy policy