Pharmacovigilance in Bulgaria
How is it with the drug safety system in one of the “Oldest European Countries”? Read our guide to get a clear picture.
And of course, if you feel you need a local consultant, we’re here for you.
Current status of our Bulgarian pharmacovigilance services:
LCPPV / EU QPPV in Bulgaria
AVAILABLE
Easily managed through Tepsivo Platform >
Local Literature Screening
AVAILABLE
Automated monitoring with Tepsivo Literature >
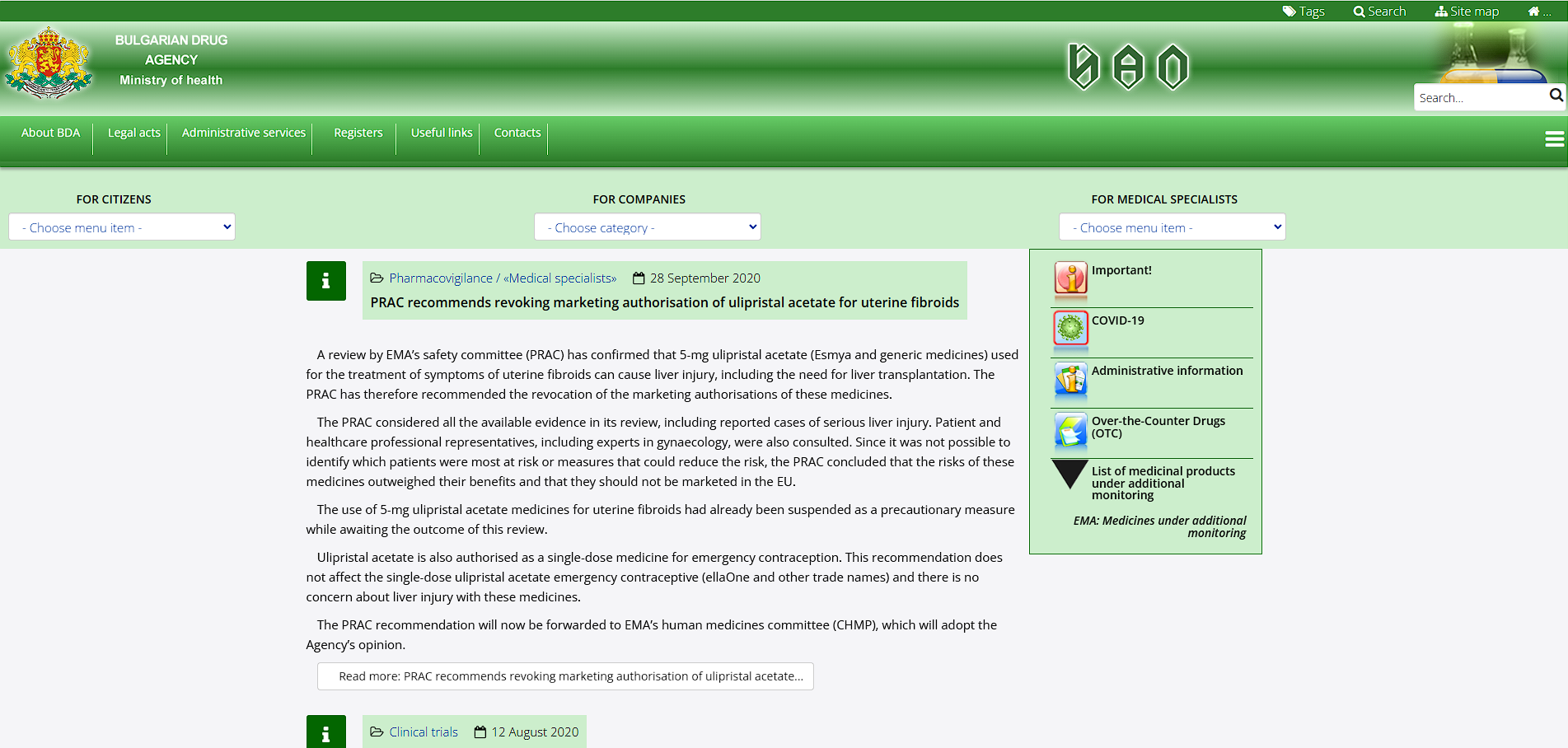
Who is the main PV authority in Bulgaria?
Other important institutions & industry groups
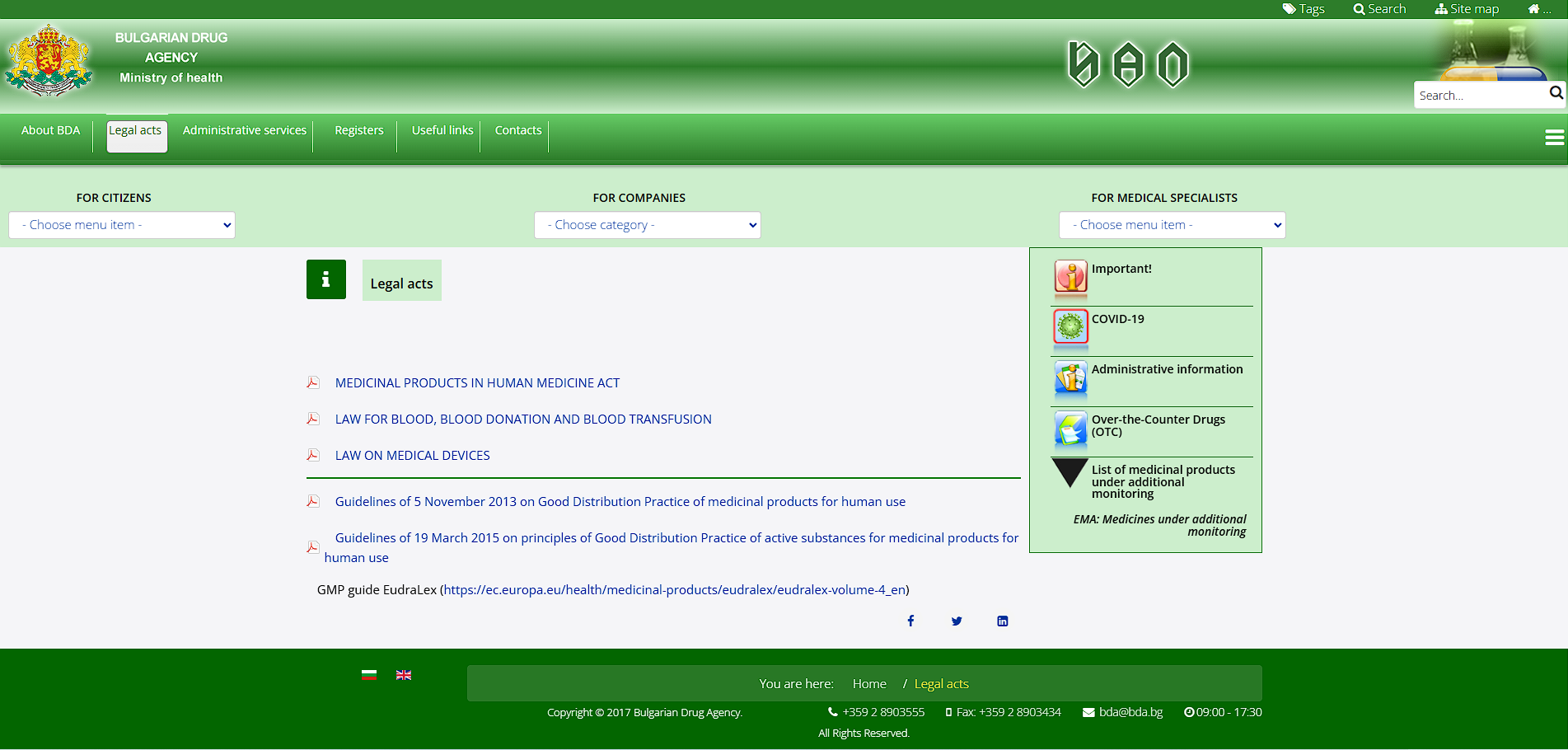
Some of the most important laws
Essential EU Legislation and EMA Guidlines
Since Bulgaria is a member of the EU, every Marketing Authorization Holder with a product on the Bulgarian market needs to follow the European pharmacovigilance legislation and guidelines by EMA (European Medicine Agency).
To navigate through the European regulations, its is useful to know following:
– EMA Pharmacovigilance System Manual
– Directive 2001/83/EC
– Regulation (EC) 726/2004
– Commission Implementing Regulation (EU) 520/2012
Local pharmacovigilance specifics
Useful abbreviations to know
If you want to study local materials about pharmacovigilance in Bulgaria, following terms will be helpful for you to get familiar with:
EMA (European Medicine Organization)
→ Key part of the EU, dedicated to ensuring the safety and effectiveness of medicines.
EVPM (EudraVigilance post-authorization module)
→ Dedicated to the collection of ICSRs related to all medicinal products authorised in the EEA.
PRAC (Pharmacovigilance Risk Assessment Committee)
→ Committee responsible for assessing and monitoring the safety of human medicines
BDA (Изпълнителната агенция по лекарствата)
→ Bulgarian Drug Agency / main authority on pharmacovigilance
Is LCPPV needed or is EU QPPV enough?
How can be adverse events reported in Bulgaria?
Reports can be made electronically via the BDA’s reporting system (healthcare professionals & public) or directly to the MAH of the drug suspected to have caused the ADR, Marketing Authorisation Holders should report via EudraVigilance.
Local medical journals to screen
Here are few examples of Bulgarian local literature sources that medical authorization holders should monitor:
ACTA MEDICA BULGARICA (AMB)
Leading scientific medical journal published by the Medical University of Sofia since 1973.
It offers a peer-reviewed platform for original research, case reports, and reviews across various medical fields, including pharmacovigilance, making it a vital resource for healthcare professionals and researchers.
ISSN: 2719-5384
JOURNAL OF IMAB
A peer-reviewed scientific publication that covers a wide range of topics in medicine and dentistry.
It is notable for its open-access model, allowing free access to full texts, which promotes the dissemination of research findings.
ISSN: 1312-773X
BULGARIAN JOURNAL OF PUBLIC HEALTH
This journal focuses on public health issues in Bulgaria and publishes research articles that address health policies, epidemiology, and community health initiatives.
It is instrumental in promoting public health awareness and research within the country.
ISSN: 1539-3704
Rather then spending your budget for hours of repetitive manual work to monitor those sources, we recommend to simply automate this task using our Tepsivo Literature solution.
Other interesting resources
bulgaria: Country Health Profile 2023
SoHEU Bulgaria – Country Health Profile 2023
A publication “State of Health in the EU” by European Observatory on Health Systems and Policies and OECD is available here in Bulgarian-specific version.
Scientific publications regarding pharmacovigilance
Reporting of Adverse Drug Reactions by Patients in Bulgaria
This publication examines and analyzes patient participation in the pharmacovigilance system in Bulgaria, providing valuable data on how patients report ADRs. It emphasizes the importance of patient involvement in drug safety monitoring.
Discover how Bulgaria and the Republic of North Macedonia are empowering patients to report adverse drug reactions directly, enhancing the safety of medicinal products in both countries. This article explores the similarities and differences in their pharmacovigilance systems, shedding light on the vital role of patient feedback in drug safety.
Attitude of medical doctors to adverse drug reactions reporting in Bulgaria
This research uncovers a troubling gap in how medical doctors in Bulgaria perceive and report Adverse Drug Reactions (ADRs), despite recognizing their importance for patient safety. With a significant number of doctors feeling overwhelmed and undertrained, the study highlights the urgent need for better education and awareness around pharmacovigilance.
interesting resources
This page contains basic staticsits about Bulgaria in various fields such as Population, Life expectancy or Health statistics.
Do you need some help with putting the Bulgarian PV System together? We’re here for you.

And while you’re here…
…consider also our global end-to-end PV solution!
Thanks to the Tepsivo platform, you can get a full pharmacovigilance system in all countries you need – for a fraction of the usual budget.

Tepsivo Oy | Urho Kekkosen katu, 4-6 E, 00 100, Helsinki, Finland | VAT number FI31367614 | contact@tepsivo.com | +358 402 204 698 | Privacy policy