No time to read now?
-> Download the article as a handy pdf
List of contents
What is the latest with DADI?
A closer look into the updates on the project
Martti Ahtola | Sep 14, 2022

Description of DADI
The goal of the European Medicines Regulatory Network’s DADI project is to replace the current PDF-based electronic application forms with web forms. The aim of the DADI project is to make electronic application form (eAF) completion and submission to authorities more efficient.
DADI project was initially announced in March 2021. While on the surface it seems like a simple digitalization project, it is the starting point of entering or updating product information data for a medicinal product, making the new digital form highly important in the set of processes under ISO IDMP standards.
Digitalized forms will eventually apply to most of the forms currently created with the .pdf templates:
- Variations
- Initial marketing authorization applications
- Renewals (human)
Other submissions are under consideration, including human and veterinary forms for centrally authorized product (CAPs) and nationally authorized product (NAPs) applications.
To understand DADI more comprehensively and in context, check out our blog posts “What is DADI?” and “What is IDMP?”.
Revised Scope
In the beginning of July 2022, EMA published at the eSubmission website information about revised go-live scope of DADI and an updated timeline.
EMA announced that the scope of the October 2022 go-live will be limited to Centrally Authorised Products (CAPs). According to the post on the eSubmission site, the decision to revise the scope was made: “due to the complexity in synchronisation of the data between xEVMPD and PMS.”
In the October 2022 release of the form, the available data for CAPs is coming from EMA internal database and it is the same dataset currently used in IRIS to support inspections.
This version of the form cannot yet be used for applications containing Nationally Authorised Products (NAPs), including National Procedures, Mutual Recognition Procedure, and Decentralised Procedure.
New Timeline
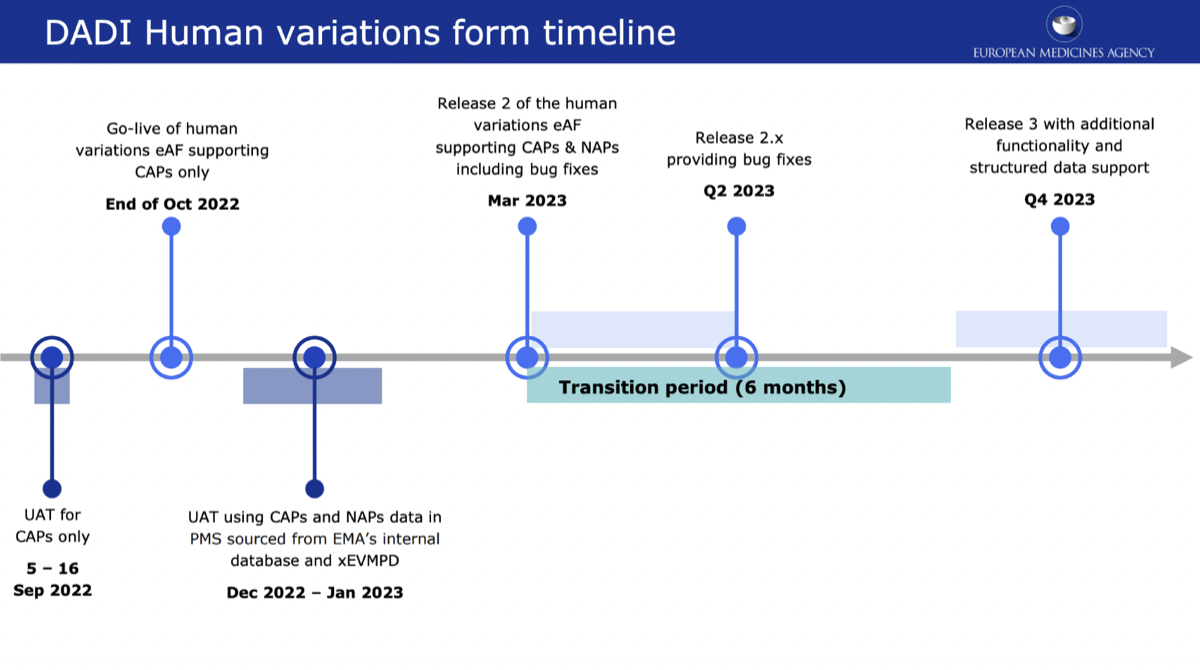
DADI UAT for CAPs is planned to be performed in September 2022. Go-live of human variations eAF (for CAPs only) is happening at the end of October 2022. The current planned timeline for releasing a version 2 of human variations eAF that supports also NAPs is March 2023 which would then start a 6-month transition period to move from the current .pdf forms completely to the electronic forms by the beginning of Q4 2023. Before that, there is a second round of UAT later in 2022 during which more MAHs and stakeholders should be able to try out the forms.
User Acceptance Testing
In July 2022, EMA also announced a call for volunteers for DADI Human Variations eAF UAT. The UAT is planned to be conducted between September 5 and September 16, 2022. A production-like version of the eAF Portal will be open for testing.
According to the EMA, the UAT participants are expected to perform testing activities, by filling in electronic Application Forms as close as possible to real-life scenarios: Filling in an eAF for one or multiple CAPs, single scope or grouping of scopes from different variation procedure types (e.g., type IA or type II), and to export the .pdf output to be included in a submission.
For some reason, EMA had decided that they would be performing this UAT but only wanted to receive a summary of the feedback from the users. EMA has delegated the feedback collection to independent industry organizations and appointed coordinators at these industry groups who are responsible for going through all the user feedback and report to the agency. These industry associations “shortlisted to collect feedback” are:
- AESGP
- EFPIA
- Europharm
- EuropaBio
- Medicines for Europe
- Vaccines Europe
What if your company is not part of these associations or their local member associations? EMA has instructed: ”if you are not a member of an Industry Association, i.e., for example, you represent a consultancy, please check with the company you represent if they can grant you access in IAM and if you can report the findings through the Industry Association they belong to.” But what if you are a marketing authorization holder who does not belong to these groups?
Tepsivo wanted to participate in the UAT, and we contacted one of our MAH customers with whom we closely collaborate with to jointly attend the UAT. However, they were not part of any of these associations either. Neither Tepsivo nor our customer were accepted to the UAT even though we suggested to perform the bug reporting through one of the organizations that offers free memberships.
As described in the legislation, EMA is supposed to perform the implementation of IDMP of which DADI is part of. It would be great if the agency performed their legally required activities that they are paid (mainly by the industry) to do.
Training
As this is a new technological thing for the agency and for the industry, there are of course several trainings and Q&A documents available. First Q&A webinar already passed but the next one will be in the beginning of September. No need to worry if you missed the registrations, the webinar recordings will be available online and surely there will be more sessions later this year or during the transition period next year.
EMA has already published a Q&A document which is definitely an interesting read into the questions that we and many others in the industry have. It is worth checking out, even if only for the comical aspects of communication from the agency that is guaranteed to give a few laughs for those who appreciate good old soul-crushing bureaucracy.
Conclusion
In conclusion, DADI is still very much a work-in-progress, but it seems that the new truly digital eAFs will be out in the wild soon and users from the pharmaceutical industry can soon start playing around with them.
There are still many questions to be answered before the digital eAF can really replace the clunky .pdf forms, but it can be seen for example from the Q&A document that the pharmaceutical community has a lot of interest and willingness to push this thing forward. This gives a good indication how much pain the current .pdf forms give to those who have to use them regularly. DADI project has the potential for making variations easier, getting rid of xEVMPD updates and in general bringing the product information management to this century.
As we warned in the IDMP post, it is better not to put too much effort in your organization’s IDMP implementation just yet even though EMA has been advising the industry to do so already for a while. The guidance for DADI and rest of IDMP implementation are still half-baked and EMA has still not figured out all the issues. It is likely to still take a while before companies can actually start using these systems.
Did you like the article? Share with your network!
…or tell us your opinion.
Follow our newsletter!
Keep up with industry trends and get interesting reads like this one 1x per month into your inbox.
Learn more about Tepsivo
We deliver modern PV solutions to fulfill your regulatory needs using less resources. See how we do it >

0 Comments