Pharmacovigilance in Portugal
How is it with the drug safety system in this state on the Iberian Peninsula? Read our guide to get a clear picture.
And of course, if you feel you need a local consultant, we’re here for you.
Current status of our Portuguese pharmacovigilance services:
LCPPV / EU QPPV in Portugal
AVAILABLE
Easily managed through Tepsivo Platform >
Local Literature Screening
AVAILABLE
Automated monitoring with Tepsivo Literature >
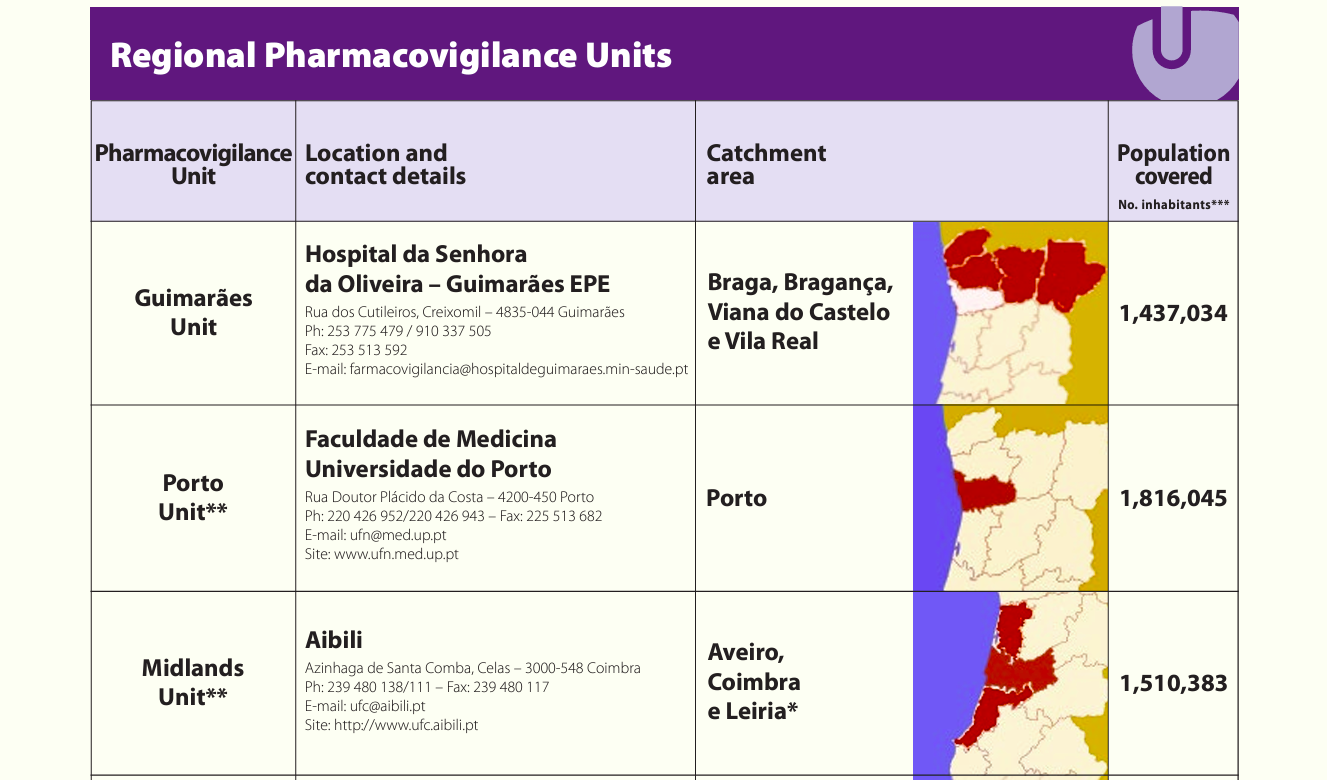
Who is the main PV authority in Portugal?
Other important institutions & industry groups
Some of the most important laws
Essential EU Legislation and EMA Guidlines
Since Portugal is a member of the EU, every Marketing Authorization Holder with a product on the Portuguese market needs to follow the European pharmacovigilance legislation and guidelines by EMA (European Medicine Agency).
To navigate through the European regulations, its is useful to know following:
– EMA Pharmacovigilance System Manual
– Directive 2001/83/EC
– Regulation (EC) 726/2004
– Commission Implementing Regulation (EU) 520/2012
Local pharmacovigilance specifics
Useful abbreviations to know
If you want to study local materials about pharmacovigilance in Portugal, following terms will be helpful for you to get familiar with:
EMA (European Medicine Organization)
→ Key part of the EU, dedicated to ensuring the safety and effectiveness of medicines.
EVPM (EudraVigilance post-authorization module)
→ Dedicated to the collection of ICSRs related to all medicinal products authorised in the EEA.
PRAC (Pharmacovigilance Risk Assessment Committee)
→ Committee responsible for assessing and monitoring the safety of human medicines
INFARMED (Instituto Nacional de Farmácia e do Medicamento)
→ Main authority on pharmacovigilance.
SNVDM (Sistema Nacional de Vigilância de Dispositivos Médicos)
→ National Medical Device Surveillance System / It aims to monitor incidents related to medical devices.
Portal RAM (Portal de notificação de suspeitas de reações adversas a medicamentos)
→ RAM Portal / Portal for reporting suspected adverse drug reactions
Is LCPPV needed or is EU QPPV enough?
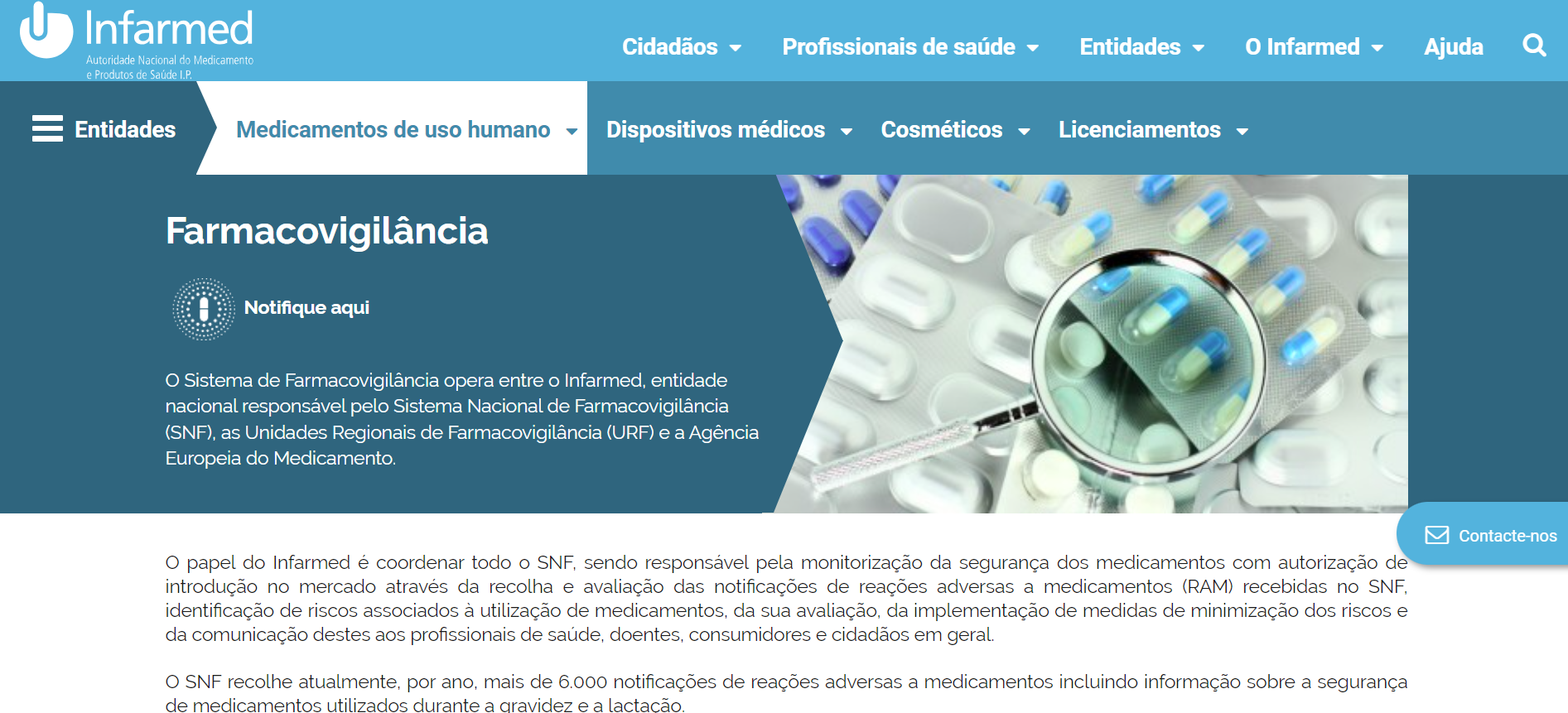
How can be adverse events reported in Portugal?
Reports can be submitted electronically through the RAM Portal, which is coordinated by INFARMED.
After clicking on the link, you’ll encourage to select whether the origin of the adverse reaction is COVID-19 vaccine or other vaccine and medicine and also whether you are a Healthcare professional or Public, more information here.
Local medical journals to screen
Here are few examples of Portuguese local literature sources that medical authorization holders should monitor:
REVISTA PORTUGUESA DE FARMACOTERAPIA
Covering latest advancements in pharmacotherapy, offering valuable insights into drug efficacy and safety, which is essential for healthcare professionals looking to enhance patient treatment strategies.
Its vast field of research articles and clinical case studies makes it a vital resource for anyone interested in improving pharmaceutical care in Portugal.
ISSN: 2183-7341
PORTUGUESE JOURNAL OF PUBLIC HEALTH
It serves as a platform for discussing public health issues and policies, featuring research that addresses the social determinants of health
This journal is particularly valuable for public health practitioners and researchers seeking to understand and improve health outcomes within the Portuguese context.
ISSN: 2504-3145
ACTA MÉDICA PORTUGUESA
It publishes original research, reviews, and clinical guidelines across a broad spectrum of medical disciplines, making it a comprehensive resource for healthcare professionals.
Its emphasis on evidence-based medicine and clinical practice ensures that readers stay informed about the latest developments in medical science relevant to Portugal.
ISSN: 1646-0758
Rather then spending your budget for hours of repetitive manual work to monitor those sources, we recommend to simply automate this task using our Tepsivo Literature solution.
Other interesting resources
Country Public Health / Health System Assessments
Portugal: Country Health Profile 2023
The 2023 edition of “State of Health in the EU: Portugal” provides a comprehensive look at the health landscape in the country, exploring key topics such as overall health status, important health determinants and risk factors, and how the health system is organized and funded.
Publications by INFARMED
As it seems the INFARMED’s annual reports have its own section on the website, but if the link stopped working we recommend to search it manually, when searching use “Relatório anual”. Here is Annual activity report 2023, only in Portuguese.
Analysis of the consumption of non-prescription medicines outside pharmacies
Infographics covering covering the prescription in Portugal since 2006, there is a PDF with statistics under each year, in Portuguese only.
Here you’ll actually find just one statistic despite the title and that is Medicines and Health Products Statistics, which is published annualy and it is accesible in both Portuguese and English.
Report: National Pharmacovigilance System 2023
By the end of 2023, the ADR Reporting Portal had documented around 155,497 reports of suspected adverse drug reactions (ADRs), with healthcare professionals being the primary contributors. This publication highlights trends in ADR reporting, emphasizing the need for increased awareness about the importance of documenting these reactions to enhance patient safety and improve healthcare outcomes.
The link for the report itself is on the linked page at the end fo the paragraph, accessible only in Portuguese.
Studies related to pharmacovigilance in Portugal
In this pharmacovigilance study, researchers investigate the incidence of spontaneously reported suspected adverse drug reactions (ADRs) associated with oral anticoagulants. By shedding light on the safety profiles of widely used medications, the study aims to enhance understanding of their risks and benefits.
In this comprehensive study, researchers analyzed 3,692 spontaneous reports of ADRs among individuals aged 65 and older, revealing that the most commonly reported ADRs fell into categories related to general disorders and skin conditions. This review sheds light on the critical need for vigilance in monitoring these drugs, making it a must-read.
This study highlights how the implementation of a newly developed active pharmacovigilance model for oral antineoplastic agents has significantly improved toxicity management and clinical outcomes for patients, fostering a supportive environment where patients feel empowered to report adverse events.
Quality of Spontaneous Reports of Adverse Drug Reactions Sent to a Regional Pharmacovigilance Unit
This study uncovers critical gaps in the documentation of adverse drug reactions (ADRs), revealing that serious ADRs are often reported incompletely, which complicates accurate causality assessments and effective risk management. Readers will find compelling evidence that highlights the urgent need for improved training on ADR reporting.
Do you need some help with putting the Portuguese PV System together? We’re here for you.

And while you’re here…
…consider also our global end-to-end PV solution!
Thanks to the Tepsivo platform, you can get a full pharmacovigilance system in all countries you need – for a fraction of the usual budget.

Tepsivo Oy | Urho Kekkosen katu, 4-6 E, 00 100, Helsinki, Finland | VAT number FI31367614 | contact@tepsivo.com | +358 402 204 698 | Privacy policy